RESEARCH AREAS

Social interactions between individuals and among groups are a hallmark of human society and are critical to the physical and mental health of a wide variety of species including humans. The central goal of our lab is to study the fundamental principles of how social behavior is regulated in the brain. We study how neural circuits and the underlying computation regulate social behavioral decisions within a single brain as well as how emergent inter-brain neural properties arise from social interactions between individuals. We take a multi-disciplinary approach and use a variety of experimental and computational technologies across molecular, circuit, and behavioral levels.
1. Neural Mechanisms of Social Information Processing and Social Decision-Making
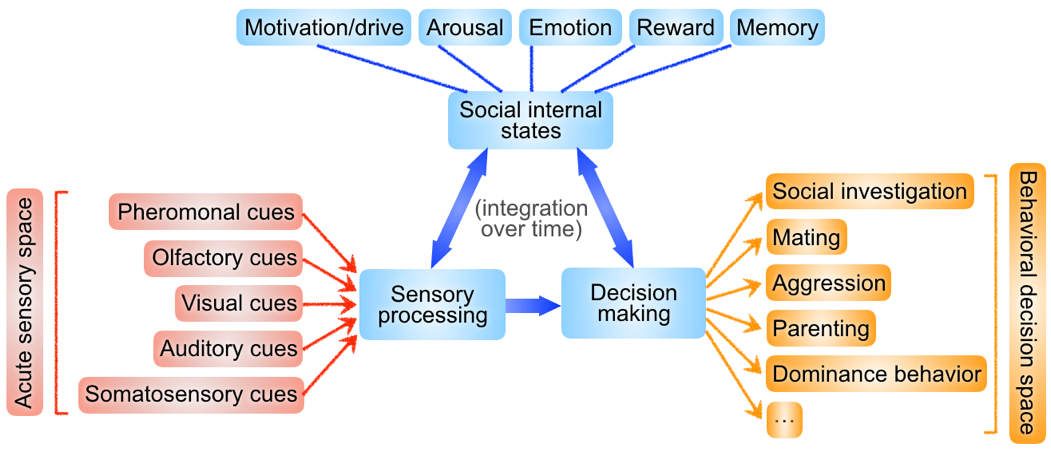
Social interactions involve the active detection of various social cues and the selection of appropriate behavioral decisions. We are interested in studying (1) how social sensory information is processed and integrated in the brain and (2) how different social behavioral decisions are selected and modulated by neural circuits.
|
Review:
Chen & Hong. Neural circuit mechanisms of social behavior. Neuron 2018. Social reward and social motivation Hu et al. An amygdala-to-hypothalamus circuit for social reward Nature Neuroscience 2021 |
Representation of social information in the brain
Kingsbury et al. Cortical representations of conspecific sex shape social behavior Neuron 2020
Sexually dimorphic control of social behavior
Chen et al. Sexually dimorphic control of parenting behavior by the medial amygdala. Cell 2019
Neural mechanisms of aggressive behavior
Hong et al. Antagonistic control of social versus repetitive self-grooming behaviors by separable amygdala neuronal subsets. Cell 2014
Kingsbury et al. Cortical representations of conspecific sex shape social behavior Neuron 2020
Sexually dimorphic control of social behavior
Chen et al. Sexually dimorphic control of parenting behavior by the medial amygdala. Cell 2019
Neural mechanisms of aggressive behavior
Hong et al. Antagonistic control of social versus repetitive self-grooming behaviors by separable amygdala neuronal subsets. Cell 2014
2. The Neuroscience of Empathy, Compassion, and Prosocial Behavior
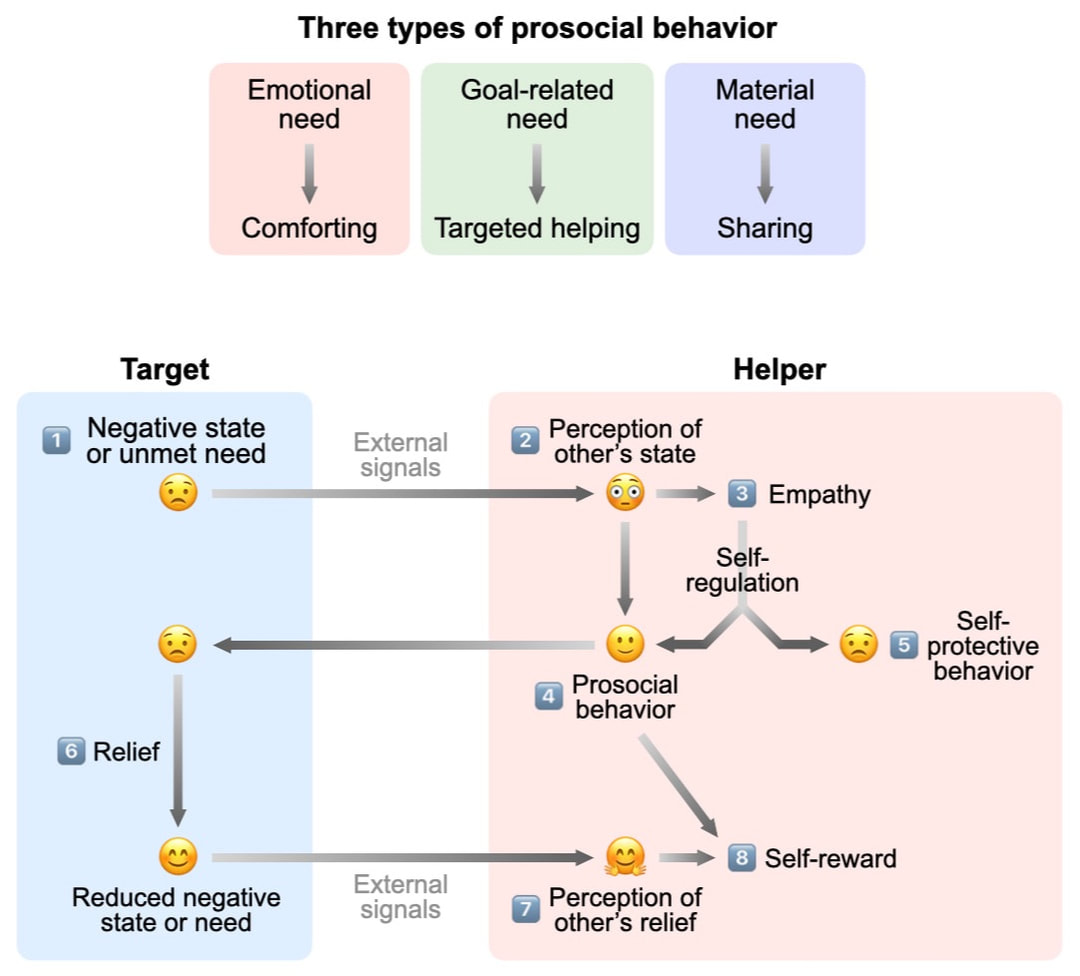
Positive, affiliative social interactions enable humans and animals to form and strengthen social bonds and build companionships, and play a crucial role in the mental and physical well-being of social species including humans. In particular, the ability to share others’ emotions and behave on others’ behalf is among the most celebrated human characteristics and is a powerful force that enhances social cohesiveness. Investigation of the neural mechanisms underlying positive social interactions may contribute to a deeper understanding of the biological basis of human generosity and kindness. We are interested in understanding how individuals display different forms of prosocial behavior and how these behaviors are regulated by neural circuits in the brain.
|
Review: Wu & Hong. Neural basis of prosocial behavior. Trends in Neurosciences 2022 Neural mechanism of helping behavior Zhang et al. Cortical regulation of helping behavior towards others in pain. Nature 2024 Neural mechanism of comforting behavior Wu et al. Neural control of affiliative touch in prosocial interaction. Nature 2021 |
3. A Multi-Brain Framework for Social Interaction
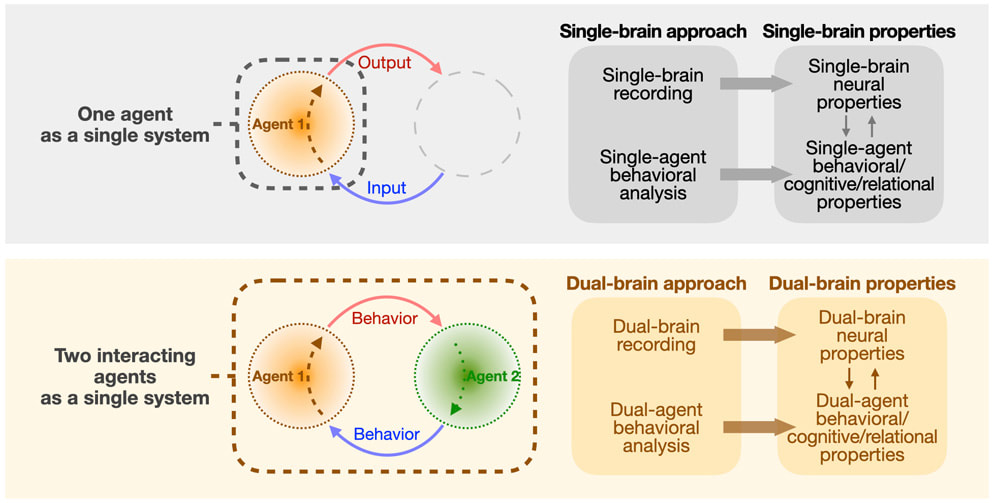
Social interaction can be seen as a dynamic feedback loop that couples two or more high-dimension neural networks (i.e. brains). This feedback loop dynamically shapes behavior, shared cognitive states, and social relationships across individual agents. A fuller understanding of the social brain requires a description of how the neural dynamics are coupled across brains and how they coevolve over time. We study social decisions and emergent inter-brain neural properties in a multi-brain framework that considers social interaction as an integrated network of neural systems.
|
Review: Kingsbury & Hong. A multi-brain framework for social interaction. Trends in Neurosciences 2020 Inter-brain neural synchrony across brains of socially interacting animals Kingsbury et al. Correlated neural activity and encoding of behavior across brains of socially interacting animals. Cell 2019 |
4. New Molecular and Behavioral Techniques for Studying Social Behavior
|
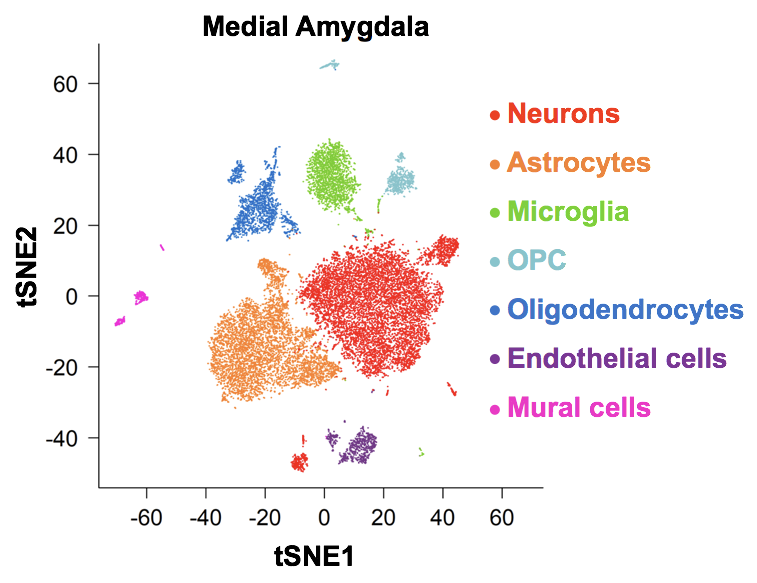
Act-seq: a single-cell sequencing approach to identify active neuronal populations
Wu et al. Detecting Activated Cell Populations Using Single-Cell RNA-Seq. Neuron 2017 Using depth sensing and machine learning to track and analyze social behavior Hong et al. Automated Measurement of Mouse Social Behaviors Using Depth Sensing, Video Tracking, and Machine Learning. Proc. Natl. Acad. Sci. USA. 2015 |